Week 3: Acids and Bases
(Click here to download the supplies and activities list)
Some substances are classified as either an acid or a base. Think of acids and bases as opposites - acids and bases are on opposite sides of a teeter-totter.
1. Hidden Messages - Goldenrod Paper
The term goldenrod is typically used to describe a color of paper - golden yellow. Certain brands of goldenrod paper contain a special dye that turns bright red in solutions that are basic like ammonia water or washing soda. The paper turns back yellow with an acid like vinegar or lemon juice. Learn how to use this special color-changing paper to develop a hidden message.
Materials
- Goldenrod paper (SteveSpanglerScience.com)(cut paper in half so that each student will have two separate pieces)
- Pencil brush
- Cotton balls
- Plastic cups
- Ammonia-water solution (Windex)
- Safety glasses
- Candle (white birthday candles)
Experiment
- Place a piece of goldenrod paper on the table. Make sure that table is clean and the work surface is dry.
- Place a drop of water on one of the corners of the paper. Does anything happen?
- Fill a jar with a small amount of ammonia water. Dip a brush in the ammonia water and "paint" with it on the goldenrod paper. Does anything happen?
- As you continue to make designs on the goldenrod paper, notice that the paper does not stay red forever. What is causing the paper to change back to yellow?
- Fill a jar with a small amount of vinegar and try to "erase" your drawing using a cotton ball or brush.
- Use a wax candle to write a secret message (such as “Hi!” or “WOW”) across the second piece of paper.
- Wipe a cotton ball with ammonia water across the secret message to see what develops.
How does it work?
The ammonia on the brush is a base and causes the dye in the special goldenrod paper to change color. You probably noticed that the red color fades over time and the paper eventually changes back to its original yellow color. Why? The carbon dioxide gas that is in the air we breathe is slightly on the acidic side of the pH scale. The carbon dioxide reacts with the ammonia on the paper to produce ammonium carbonate, which changes the pH of the paper to neutral (roughly a pH of 7) and the dye changes back yellow. If you use a stronger base like washing soda, the red message will not disappear with just the carbon dioxide in the air. You will need to use a stronger acid like lemon juice or vinegar to change it from red to yellow. You can also use goldenrod paper as inexpensive pH paper to classify safe household products as being either acidic or basic.
2. Red Cabbage Chemistry
Scientists
can tell if a substance is an acid or a base by means of an indicator.
An indicator is typically a chemical that changes color if it comes in
contact with an acid or a base. This experiment will teach you how to
make your own indicator using cabbage juice.
Materials
* Red cabbage
* Blender
* Strainer
* Clear drinking glasses
Ideas for Test chemicals: Vinegar, Windex, Baking soda, Lemon juice, Washing soda, Laundry detergent, Soda pop, Alka-Seltzer
Let’s make an indicator out of red cabbage. Here are the steps...
Experiment
- Peel off six big cabbage leaves and put them in a blender filled half full with water. Liquify it!
- Pour the purplish cabbage liquid through a strainer to filter out all of the big chunks of cabbage. Doesn’t cabbage juice smell great?
- Save the liquid for the experiments to follow.
- Set out three or more glasses, side by side in a row, against a white piece of paper. Fill each glass half full with cabbage juice.
- Since
you know that vinegar is an example of an acid, add a little vinegar to
the first glass of cabbage juice. Stir with a spoon and notice the
color change to red, which indicates that vinegar is classified as an
acid.
- In the glass furthest away from the glass to which you added vinegar add a little windex. Notice how the liquid turns green which indicates
this chemical is a base.
- Keep these two glasses of red and green liquid for future reference.
- Try
adding your own “test” substances to a small amount of cabbage juice
and note the color change to determine if something is an acid or a
base.
How does it work?
As
you can see, the purple cabbage juice turns red when it is mixed with
something acidic (an acid), or green when it mixes with something basic
(a base). In the previous experiment, the vinegar was the acid and the windex (=ammonia) was the base. Remember that an acid is the opposite
of a base. Red cabbage juice is considered to be an indicator because
it shows us something about the chemical composition of other
substances. This is just one of many indicators that are available to
scientists. Some indicators start out colorless and turn blue or pink,
for example, when they mix with a base. There are hundreds of different
types of indicators available to scientists depending on the type of
substance that they are testing. If there is no color change at all,
the substance that you are testing is probably neutral, just like water.
Use your cabbage juice indicator to test the acid or base properties of other common substances: Orange juice, lemonade, milk, salt, ammonia, or soap are some suggestions. You’ve heard about how acid rain is destroying the environment. Collect a sample of rain water and test its acidity using your cabbage indicator. These are all great variables to test if you want to make Red Cabbage Chemistry an awesome Science Fair project.
3. pH paper
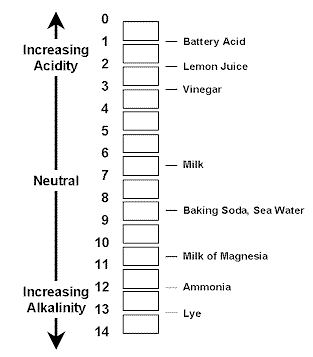
The pH of a substance is a measure of how acidic or alkaline a substance is (alkaline is another word for basic).
Acids have a pH value between 0 and 7
Alkalines have a pH value between 7 and 14
(if a substance has a pH value of 7, it is neutral – neither acidic or alkaline)
A pH can be measured by dipping a pH paper into solutions such as water and other substances.
PH paper is also known as litmus paper, this a special type of paper containing a chemical that will tell you the pH of a substance by the color it turns when it is dipped into the substance.
For this experiment dip pH paper into the different solutions provided and write down on your pH scale where the solutions fall on the scale. Is the solution you tested an acid or a base?